I have spoken many times about the role of a clinical diagnosis of COVID-19 (cough, fever, sweats, and changes in taste or smell) which is free, repeatable, does not require a doctor’s order, can be done by a third-grader versus the expensive, one-time, slow nasal swab test for viral genetic material.
A group of scientists from none other than Johns Hopkins have confirmed my suspicions and, in fact, the test is worse than I thought. The key parameter that this test needs to have to beat my simple clinical test is a low false negative rate. After all, what some people say they need is a test that works very soon after an infection begins and then is positive for as long as a patient is shedding virus and can potentially infect others. This parameter is called the ‘False Negative’ rate and is the percent of patients who really have an infection with SARS-CoV-2 but who test negative with the nasal swab test.
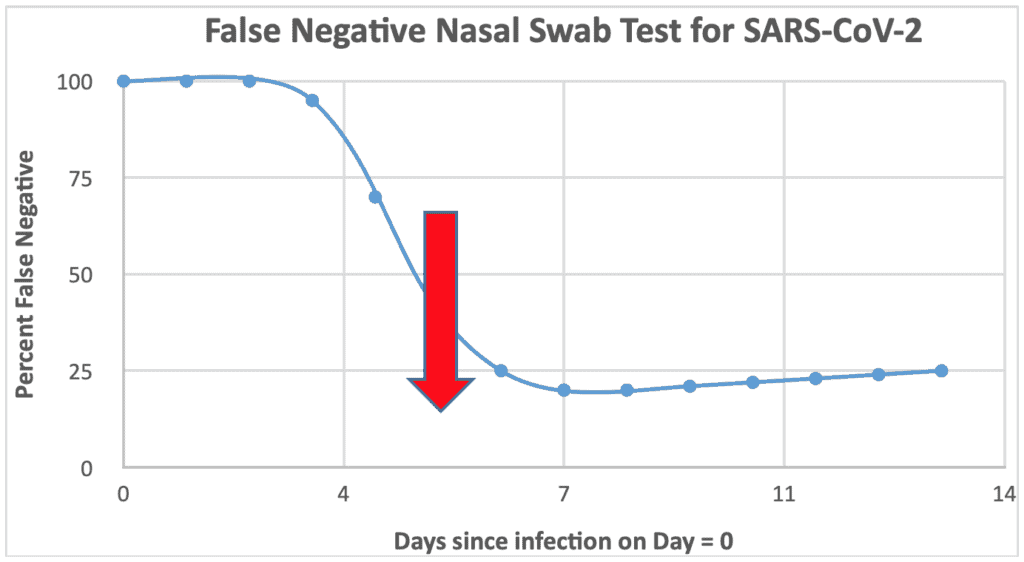
The results are below and, as I said, are worse than I suspected. The graph starts at 100% False Negative on the day of infection. After all it is just the moment a person is in a room, train, plane, bar, etc. and takes a breath just as someone with COVID-19 sneezes. And for the next three days, as the infection silently builds in the nose and throat, the nasal swab test stays almost completely negative, but falsely so since the person here is really infected. Day 4 and 5 show us the test is finally working, with the False Negative rate dropping to about 70% and 40%, respectively.
But wait! What is that big red arrow on Day 5? That is the clinical symptoms of fever, cough, sweats, and changes in taste or smell finally showing up. Great you say: I have this high tech test that is no better than a third grader would do. Once the symptoms set in the test runs about a 20% False Negative Rate for the rest of the infection. Isn’t it interesting that the clinical symptom test also shows about 20% asymptomatic patients? So don’t let someone tell you that we need tests to pick up all the asymptomatic cases. (Although I suspect if you had infinite dollars you could do both and probably get the False Negative rate down to 4% or so; thinking 0.2 times 0.2 equals 0.04 if they are independent but since they probably both happen in patients with low virus loads, they will be connected and miss the same patients, so maybe 10% False Negatives).
If this summary isn’t enough for you and you want to dig in deep, the full article can be found at this link. (1)
My COVID-19 Book Publishes June 5, 2020 on Amazon Kindle!
My book entitled, COVID-19 Survival Manual: A Physician’s Guide to Keep You and Your Family Healthy During the Pandemic and Beyond, will be published as an Amazon Kindle book on June 5, 2020, and will provide my blueprint for how to move beyond this pandemic safely for you and your family. Before anyone decides to return to life outside the shelter, please read my book. It might save your life!
COVID-19 Survival Manual: A Physician’s Guide to Keep You and Your Family Healthy during the Pandemic and Beyond
This book is chock-full of lifesaving tips you can’t find anywhere else to help you and your family stay safe while sheltering-in-place, as well as how to get back to life in the coming weeks and months.
Some highlights:
• One 2-minute step you can take every time you come home to kill Coronavirus before it enters your lungs
• A, quick, free home test for COVID-19 that is as good as the FDA-approved nasal swab
• The one exercise you won’t learn in the gym that can save your life
• How NOT to die of COVID-19 (Make your lungs younger)
• What to do if you have early stage COVID-19 so that you can stay out of the hospital
• The #1 most effective way to prevent the spread of coronavirus as we reopen society (Hint: it’s the one thing the CDC said was “NOT effective” when coronavirus hit the U.S.)
• Why a vaccine won’t be the solution, and what YOU can do to protect yourself, now and in the future
• What to eat and drink (and what to avoid) so you can prevent and beat this coronavirus
• The best supplements I have found from clinical trial research for immune health during this pandemic
• The coronavirus’ “Diabolic Trait” and how it helped the virus spread so fast
• An easy DIY step that takes your face mask from a viral barrier to a virus killer, giving you over 100-times the protection of an untreated mask
• The one blood test to ask for if you are hospitalized that can keep you off a ventilator
• Three steps to take to thrive during the next epidemic
• What is gain-of-function research and why we must ban it to prevent future pandemics
It will be available on Amazon Kindle for $4.99 starting June 5. The proceeds will be shared with Team Rubicon, a non-profit organization that serves communities by mobilizing veterans to continue their service, leveraging their skills and experience to help people prepare, respond, and recover from disasters and humanitarian crises.
You can get more information on my Amazon author page: www.amazon.com/author/stevenquay




